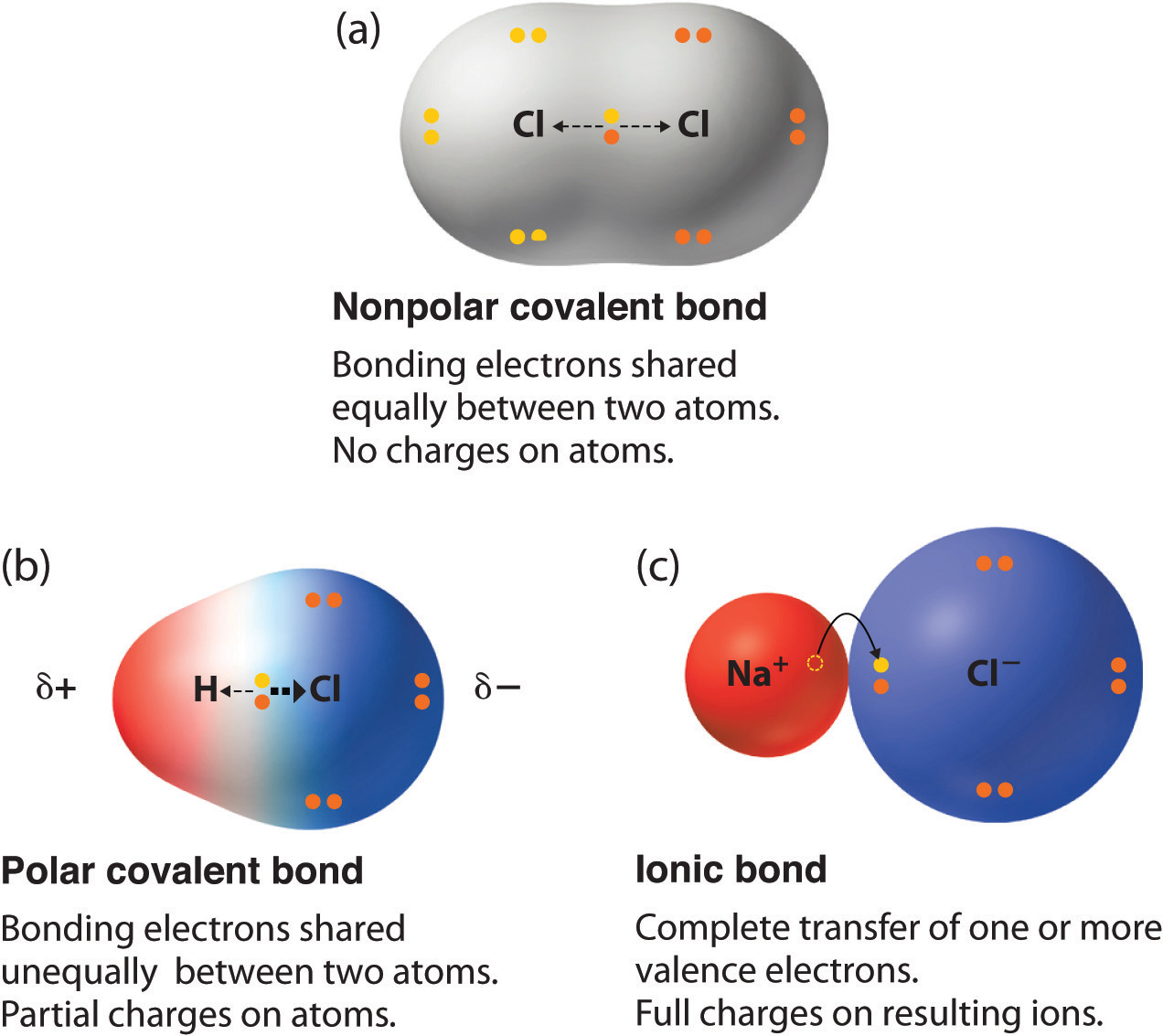
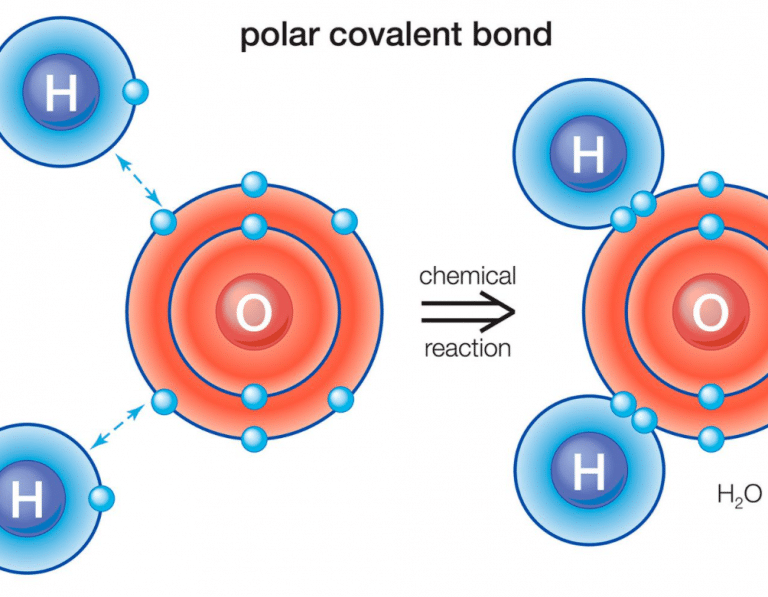
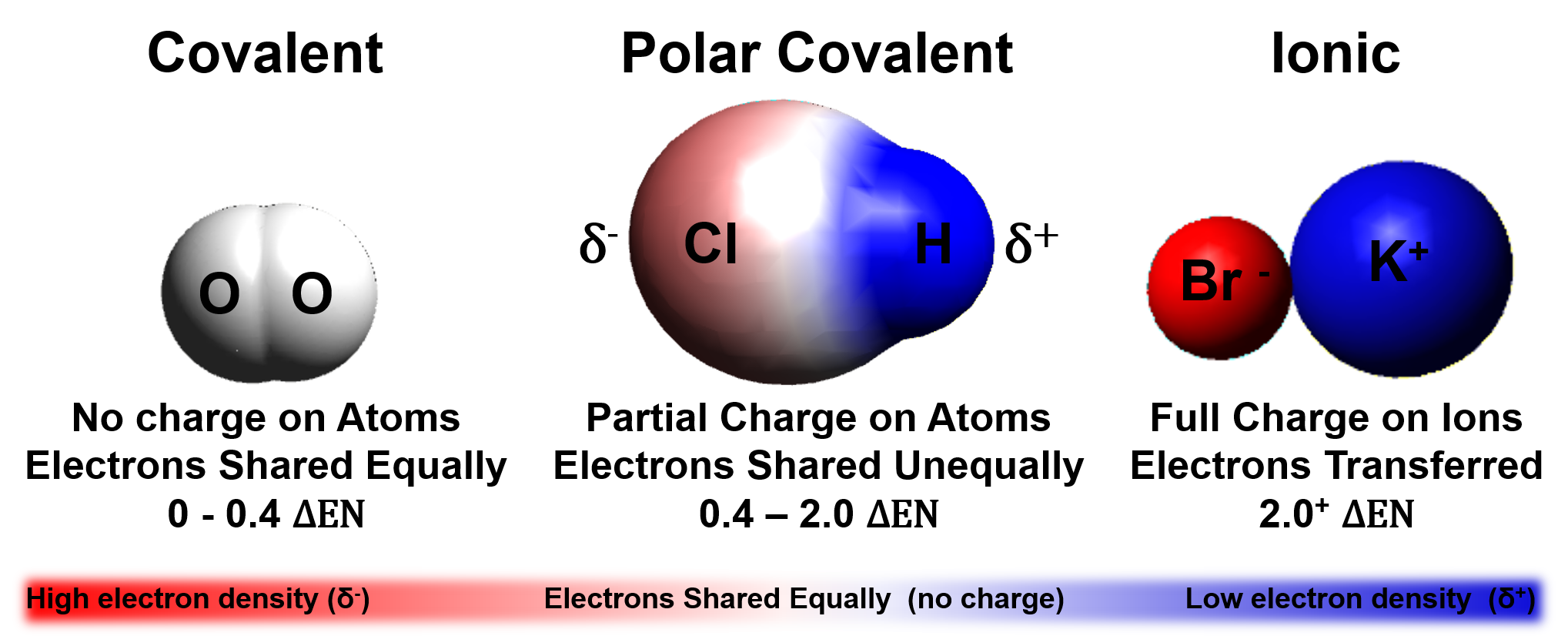
Which Combination Of Atoms Can Form A Polar Covalent Bond
Which Combination Of Atoms Can Form A Polar Covalent Bond - Web non polar covalent bond is defined as type of chemical bond with equal sharing of the bond electron rises when the electronegativity of the two atoms are equal. Let us consider a and b in which them is electronegativity difference. Web it takes two electrons to make a covalent bond, one from each bonding atom. The common factors that affect the conjugate base's stability are 1) the size and. Web which combination of atoms can form a polar covalent bond? Two atoms with equal electronegativity will. Web this creates a spectrum of polarity, with ionic (polar) at one extreme, covalent (nonpolar) at another, and polar covalent in the middle. Web covalent bonds are also affected by the electronegativity of the connected atoms which determines the chemical polarity of the bond. Web some compounds contain both covalent and ionic bonds. The polarity of a bond depends on the electronegativities of the bonded atoms.
Web advanced physics advanced physics questions and answers which combination of atoms can form a polar covalent bond? A) hand br ob) hand h c) na and br od) n. Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally. Web 1)molecular compounds where in atoms are joined by covalent bonds. Web what kind of bond is formed when two atoms share electrons to form a molecule? Web the acid that forms the more stable conjugate base will be the stronger acid. Two atoms with equal electronegativity will. Web it takes two electrons to make a covalent bond, one from each bonding atom. Web bonds between carbon and other elements such as oxygen and nitrogen are polar. Web which combination of atoms can form a polar covalent bond?
Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally. This is due to one of the elements having a higher electronegativity than the. Only h and br form polar covalent bond. Web this creates a spectrum of polarity, with ionic (polar) at one extreme, covalent (nonpolar) at another, and polar covalent in the middle. 2) ionic compounds where atoms are joined by ionic bond. The atoms in this bond are xenon (electronegativity 2.6) and fluoride (electronegativity 4.0). A) h and br explanation: Web polar covalent bonds are usually formed between two nonmetal atoms having different electronegativities. Web which combination of atoms can form a polar covalent bond? The polarity of a bond depends on the electronegativities of the bonded atoms.
Covalent Bonds Biology for NonMajors I
The atoms in this bond are xenon (electronegativity 2.6) and fluoride (electronegativity 4.0). A) hand br ob) hand h c) na and br od) n. In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two. This is due to one of the elements having a higher electronegativity than the. Web covalent bonds.
2.2 Chemical Bonds Anatomy & Physiology
A h and h b h and f c n and n d na and f medium solution verified by toppr correct option is b) answer= h. Two atoms with equal electronegativity will. Web what kind of bond is formed when two atoms share electrons to form a molecule? 2) ionic compounds where atoms are joined by ionic bond. Web.
How does a polar bond differ from a covalent bond
Web some compounds contain both covalent and ionic bonds. Web non polar covalent bond is defined as type of chemical bond with equal sharing of the bond electron rises when the electronegativity of the two atoms are equal. In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two. Web what kind of.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
Two atoms with equal electronegativity will. A) h and br explanation: A) hand br ob) hand h c) na and br od) n. Web 1)molecular compounds where in atoms are joined by covalent bonds. In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two.
What Is a Polar Bond? Definition and Examples
Which molecule contains a non polar covalent bond? A h and h b h and f c n and n d na and f medium solution verified by toppr correct option is b) answer= h. Two atoms with equal electronegativity will. Which type of bond is present. In a covalent bond, the stability of the bond comes from the shared.
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts
Web this creates a spectrum of polarity, with ionic (polar) at one extreme, covalent (nonpolar) at another, and polar covalent in the middle. This is due to one of the elements having a higher electronegativity than the. The common factors that affect the conjugate base's stability are 1) the size and. Web bonds between carbon and other elements such as.
Ch4 Polar Or Nonpolar Covalent Bond Which statement explains why a
In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two. Web what kind of bond is formed when two atoms share electrons to form a molecule? Web which combination of atoms can form a polar covalent bond? Web covalent bonds in which the sharing of the electron pair is unequal, with the.
Polar Covalent Bond Definitions, Types and Examples
Web some compounds contain both covalent and ionic bonds. A) h and br explanation: Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally. Only h and br form polar covalent bond. Web polar covalent bonds are usually formed between two nonmetal atoms having different electronegativities.
This figure shows the structure of a water molecule. The top panel
Web advanced physics advanced physics questions and answers which combination of atoms can form a polar covalent bond? Web non polar covalent bond is defined as type of chemical bond with equal sharing of the bond electron rises when the electronegativity of the two atoms are equal. In a covalent bond, the stability of the bond comes from the shared.
Building the World Be Prepared! Everything you should know for 1st
This is due to one of the elements having a higher electronegativity than the. Web polar covalent bonds are usually formed between two nonmetal atoms having different electronegativities. Web advanced physics advanced physics questions and answers which combination of atoms can form a polar covalent bond? Only h and br form polar covalent bond. Which molecule contains a non polar.
Web A Covalent Bond Is Formed When Two Atoms Share Electron Pairs.
Web 1)molecular compounds where in atoms are joined by covalent bonds. Web it takes two electrons to make a covalent bond, one from each bonding atom. Web some compounds contain both covalent and ionic bonds. In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two.
Web This Creates A Spectrum Of Polarity, With Ionic (Polar) At One Extreme, Covalent (Nonpolar) At Another, And Polar Covalent In The Middle.
A) hand br ob) hand h c) na and br od) n. Web non polar covalent bond is defined as type of chemical bond with equal sharing of the bond electron rises when the electronegativity of the two atoms are equal. Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally. Web which combination of atoms can form a polar covalent bond?
Web Bonds Between Carbon And Other Elements Such As Oxygen And Nitrogen Are Polar.
Web what kind of bond is formed when two atoms share electrons to form a molecule? Web the acid that forms the more stable conjugate base will be the stronger acid. Web advanced physics advanced physics questions and answers which combination of atoms can form a polar covalent bond? Web polar covalent bonds are usually formed between two nonmetal atoms having different electronegativities.
The Atoms In This Bond Are Xenon (Electronegativity 2.6) And Fluoride (Electronegativity 4.0).
Web some compounds contain both covalent and ionic bonds. This is due to one of the elements having a higher electronegativity than the. 2) ionic compounds where atoms are joined by ionic bond. A) h and br explanation:
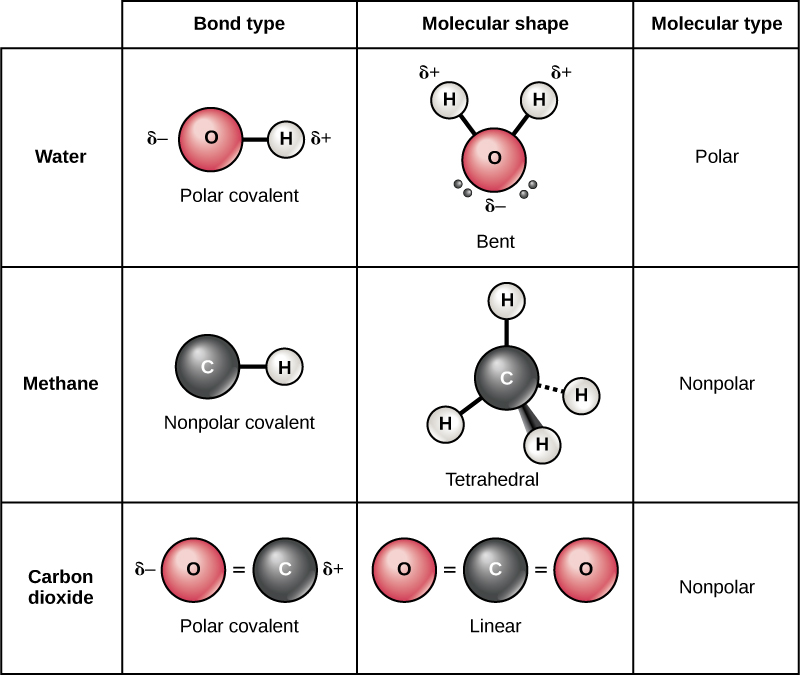

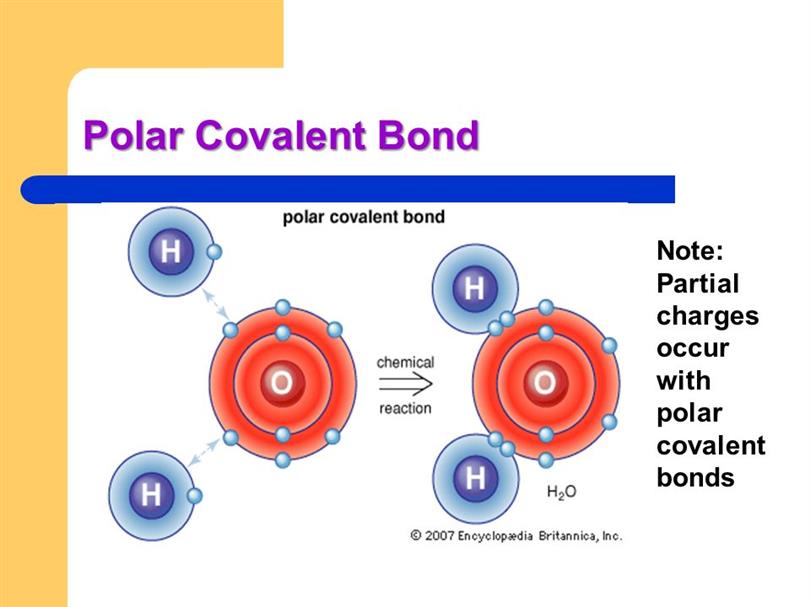
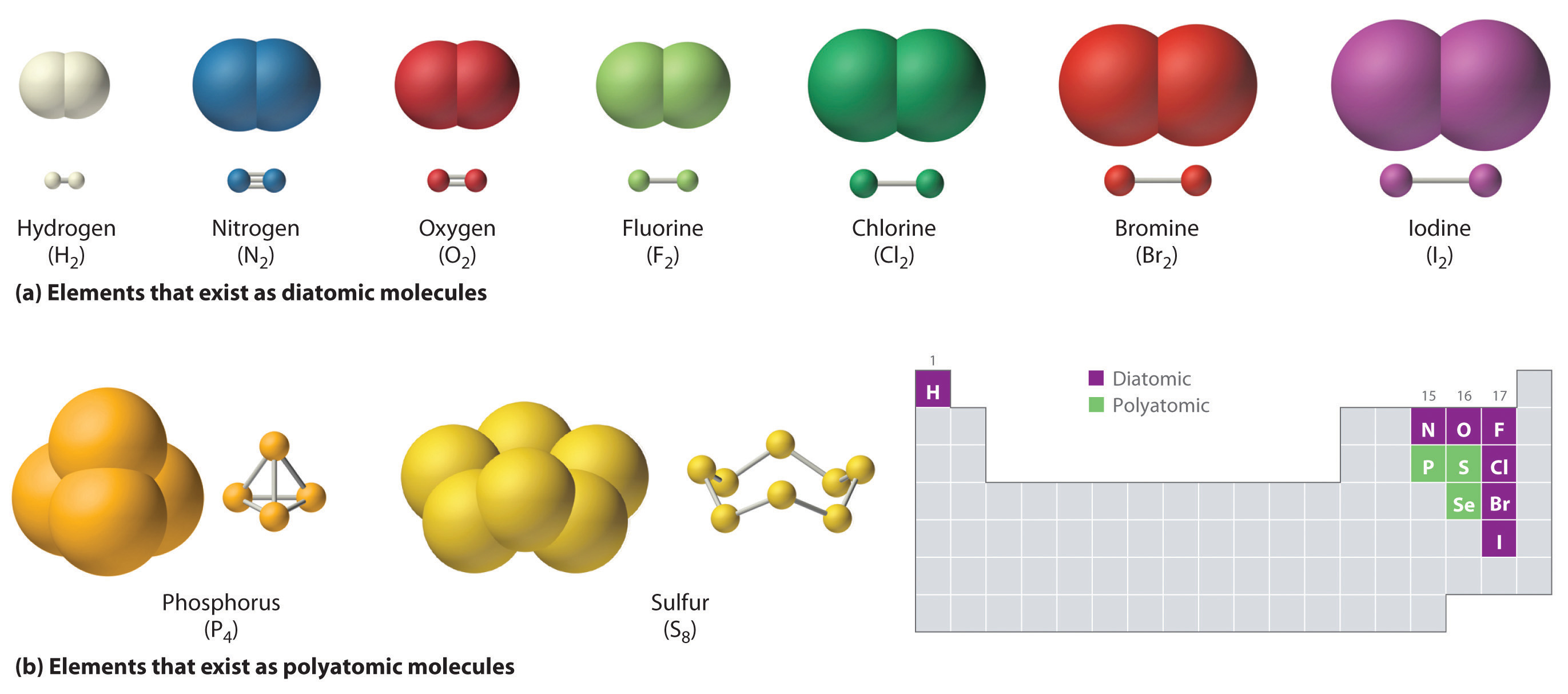
/PolarConvalentBond-58a715be3df78c345b77b57d.jpg)