How Many Covalent Bonds Does Phosphorus Form
How Many Covalent Bonds Does Phosphorus Form - Web some compounds contain both covalent and ionic bonds. So, phosphorous can exceed its valency from +3 to +5. This is summarized in the table below. Group 5a form 3 bonds; Web how many covalent bonds does a phosphorus atom normally form, based on the number of valence electrons in the atom? Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet. Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell. Group 6a form 2 bonds; Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a variety of oxidation states, from −3 to 4,.
Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet. Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell. Group 5a form 3 bonds; This is summarized in the table below. And like a central silicon atom, there are four pairs of electrons being shared with. Web how many bonds does phosphorus typically make? Web correct option is b) phosphorous has an atomic number of 15 and electronic configuration as 1s 22s 22p 63s 23p 3. Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a variety of oxidation states, from −3 to 4,. Web the number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. So there are three covalent bonds and one lone pair of electrons on.
Covalent bond two atoms form a covalent chemical bond. How many bonds does phosphorus typically make? Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet. Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. So there are three covalent bonds and one lone pair of electrons on. Web how many covalent bonds does a phosphorus atom normally form, based on the number of valence electrons in the atom? Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a variety of oxidation states, from −3 to 4,. Web the number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. So there are three covalent bonds and one lone pair of electrons on.
LabXchange
Web there are many different modifications of phosphorus in nature. So there are three covalent bonds and one lone pair of electrons on. Therefore phosphorus maximum covalency of 6. Web the formula of the carbonate ion is co 32−. Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet.
Is SiO2 Ionic or Covalent? Techiescientist
Web correct option is b) phosphorous has an atomic number of 15 and electronic configuration as 1s 22s 22p 63s 23p 3. Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet. The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as.
Atoms, Isotopes, Ions, and Molecules The Building Blocks · Biology
Web typically, the atoms of group 4a form 4 covalent bonds; So there are three covalent bonds and one lone pair of electrons on. And like a central silicon atom, there are four pairs of electrons being shared with. Web correct option is b) phosphorous has an atomic number of 15 and electronic configuration as 1s 22s 22p 63s 23p.
How Many Single Bonds Can Carbon Form fredhughesdesign
Web how many covalent bonds does a phosphorus atom normally form, based on the number of valence electrons in the atom? Web the formula of the carbonate ion is co 32−. Web the number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. The atoms of a polyatomic ion are tightly bonded.
How does phosphorus form 5 covalent bonds? The Unconditional Guru
Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell. Therefore phosphorus maximum covalency of 6. Web correct option is b) phosphorous has an atomic number of 15 and electronic configuration as 1s 22s 22p 63s 23p 3. Group 6a form 2 bonds; So, phosphorous can exceed its valency from +3 to.
__TOP__ How Many Covalent Bonds Can Chlorine Form
And like a central silicon atom, there are four pairs of electrons being shared with. Web the number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell. Web how many bonds does phosphorus typically.
Solved H2S CIOC HSH 1 How many bonds does oxygen
The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. Web the number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a.
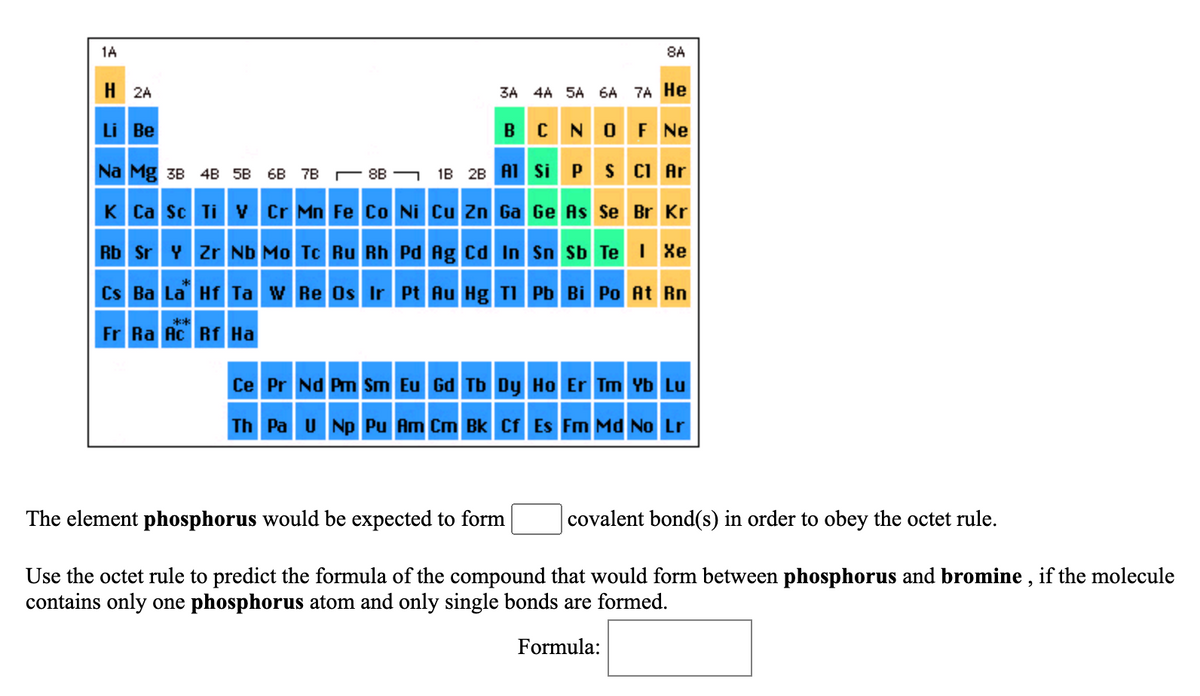
Answered The element phosphorus would be… bartleby
Web how many covalent bonds does a phosphorus atom normally form, based on the number of valence electrons in the atom? Web the formula of the carbonate ion is co 32−. Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a variety of oxidation states, from −3 to 4,. Web.
Lewis Dot Diagram Phosphorus
Group 5a form 3 bonds; Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. How many bonds does phosphorus typically make? Therefore phosphorus maximum covalency of 6. Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell.
FileElectron shell 015 Phosphorus.svg Wikimedia Commons Phosphorus
Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. Web some compounds contain both covalent and ionic bonds. Web the formula of the carbonate ion is co 32−. Web correct option is b) phosphorous has an atomic number of 15 and electronic configuration as 1s 22s 22p 63s 23p 3. And group 7a.
So There Are Three Covalent Bonds And One Lone Pair Of Electrons On.
How many bonds does phosphorus typically make? Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus. So there are three covalent bonds and one lone pair of electrons on. Web there are many different modifications of phosphorus in nature.
Web Correct Option Is B) Phosphorous Has An Atomic Number Of 15 And Electronic Configuration As 1S 22S 22P 63S 23P 3.
And group 7a form one bond. Covalent bond two atoms form a covalent chemical bond. Web the most commonly observed (formal) oxidation state of phosphorus in molecules and crystals is +3 and +5, although a variety of oxidation states, from −3 to 4,. Web the number of valence electrons in phosphorus is generally 5 and it requires 3 more electrons to complete its octet.
So, Phosphorous Can Exceed Its Valency From +3 To +5.
Web from the diagram, we can see that phosphorus has a full eight electrons in its outermost shell. Web how many bonds does phosphorus typically make? And like a central silicon atom, there are four pairs of electrons being shared with. Web in the pcl3 molecule, each chlorine is bonded by a single covalent bond with phosphorus.
Therefore Phosphorus Maximum Covalency Of 6.
The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. Web typically, the atoms of group 4a form 4 covalent bonds; This is summarized in the table below. Web how many covalent bonds does a phosphorus atom normally form, based on the number of valence electrons in the atom?