Aldehydes May Oxidize To Form
Aldehydes May Oxidize To Form - Because the solution is alkaline, the aldehyde itself is oxidized to a salt of the corresponding carboxylic acid. Web the oxidation of an alcohol to form an aldehyde or ketone is very important in synthesis. O c h o c o h oxidation ¥alcohols. Web the oxidation of aldehydes by o2 appears to be a chain mechanism involving free radicals and yielding a rather reactive peracid, which then reacts with the. Web ¥carbonyl groups in aldehydes and ketones may be oxidized to form compounds at the next òoxidation level ó, that of carboxylic acids. Web aldehydes reduce the diamminesilver(i) ion to metallic silver. They use a strong oxidant like potassium permanganate (kmno4) Hence, option b is correct. Web oxidation of alcohols to aldehydes and ketones. Web aldehydes have a proton attached to the carbonyl carbon which can be abstracted, allowing them to be easily oxidized to form carboxylic acids.
They use a strong oxidant like potassium permanganate (kmno4) O c h o c o h oxidation ¥alcohols. Web depending on the conditions of the oxidation, aldehydes will form carboxylic acids. Ketones are not oxidised under these conditions as they lack the critical h for the elimination to occur (see. In this case, excess dichromate will further oxidize the aldehyde to a. Web oxidation of alcohols to aldehydes and ketones. Oxidation of alcohols to aldehydes is partial oxidation; Aldehydes are further oxidized to carboxylic acids. Hence, option b is correct. Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids.
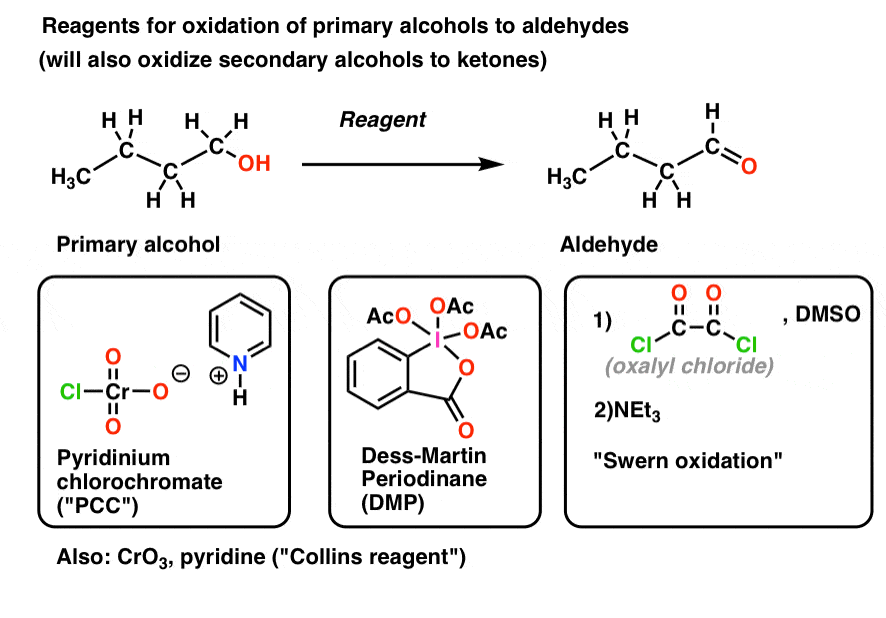
Web the product of the oxidation of an aldehyde, results in a carboxylic acid. Web aldehydes, rcho, can be oxidised to carboxylic acids, rco2h. O c h o c o h oxidation ¥alcohols. Web oxidation of 1 o alcohols to form aldehydes (section 17.7) pcc pyridinium chlorochromate ( pcc) is a milder version of chromic acid. These functional groups are useful for further reactions; Web the oxidation of an alcohol to form an aldehyde or ketone is very important in synthesis. In this case, excess dichromate will further oxidize the aldehyde to a. Aldehydes are further oxidized to carboxylic acids. Web aldehydes undergo oxidation more quickly than ketones. This will happen if the oxidation happens under acidic or alkaline conditions.
Tollens Reagent Silver Mirror Test for Aldehydes
Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids. Web currently, most investigations on aldehyde oxidations focus on aerobic oxidation, i.e., using molecular oxygen (o 2) to oxidize aldehydes into the corresponding carboxylic. Web oxidation of alcohols to aldehydes and ketones. Web ¥carbonyl groups in aldehydes and ketones may be oxidized to form compounds at the next òoxidation.
Alcohol Oxidation Mechanisms and Practice Problems Chemistry Steps
Web there are some reagents which can selectively oxidize a primary alcohol and stop at an aldehyde without complete oxidation to the acid. Web the product of the oxidation of an aldehyde, results in a carboxylic acid. Web aldehydes, rcho, can be oxidised to carboxylic acids, rco2h. Web answer 1 aldehydes are a class of organic compound which can be.
Representative examples of oxidation of aldehydes. Download
Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids. O c h o c o h oxidation ¥alcohols. Web the product of the oxidation of an aldehyde, results in a carboxylic acid. Aldehydes are further oxidized to carboxylic acids. These functional groups are useful for further reactions;
Alcohol Oxidation "Strong" & "Weak" Oxidants Master Organic Chemistry
Web aldehydes have a proton attached to the carbonyl carbon which can be abstracted, allowing them to be easily oxidized to form carboxylic acids. Web answer 1 aldehydes are a class of organic compound which can be oxidized to form corresponding carboxylic acid or organic acid. Web aldehydes reduce the diamminesilver(i) ion to metallic silver. In other words, aldehydes are.
PDF Télécharger aldehydes may oxidize to form Gratuit PDF
Primary alcohols can only be oxidized to form aldehydes or carboxylic acids.for instance, using chromic acid (to get a carboxylic acid), or using pcc in. Hence, option b is correct. In this case, excess dichromate will further oxidize the aldehyde to a. This will happen if the oxidation happens under acidic or alkaline conditions. They use a strong oxidant like.
Solved Be sure to answer all parts. Hydroxy aldehydes A and
Web depending on the conditions of the oxidation, aldehydes will form carboxylic acids. Web aldehydes reduce the diamminesilver(i) ion to metallic silver. Aldehydes are further oxidized to carboxylic acids. Hence, option b is correct. Web answer 1 aldehydes are a class of organic compound which can be oxidized to form corresponding carboxylic acid or organic acid.
Pathways leading to formation of oxidation and nitric oxide (NO
Web oxidation of 1 o alcohols to form aldehydes (section 17.7) pcc pyridinium chlorochromate ( pcc) is a milder version of chromic acid. Web depending on the conditions of the oxidation, aldehydes will form carboxylic acids. Ketones are not oxidised under these conditions as they lack the critical h for the elimination to occur (see. Because the solution is alkaline,.
Organic Chemistry, Form, Part 14 Aldehydes Structure and
Aldehydes are further oxidized to carboxylic acids. Web aldehydes have a proton attached to the carbonyl carbon which can be abstracted, allowing them to be easily oxidized to form carboxylic acids. Web the product of the oxidation of an aldehyde, results in a carboxylic acid. Web aldehydes, rcho, can be oxidised to carboxylic acids, rco2h. Web depending on the conditions.
PPT Aldehydes, Ketones, and carboxylic acids PowerPoint Presentation
Web oxidation of 1 o alcohols to form aldehydes (section 17.7) pcc pyridinium chlorochromate ( pcc) is a milder version of chromic acid. Web aldehydes have a proton attached to the carbonyl carbon which can be abstracted, allowing them to be easily oxidized to form carboxylic acids. In other words, aldehydes are better at reducing than ketones due to the.
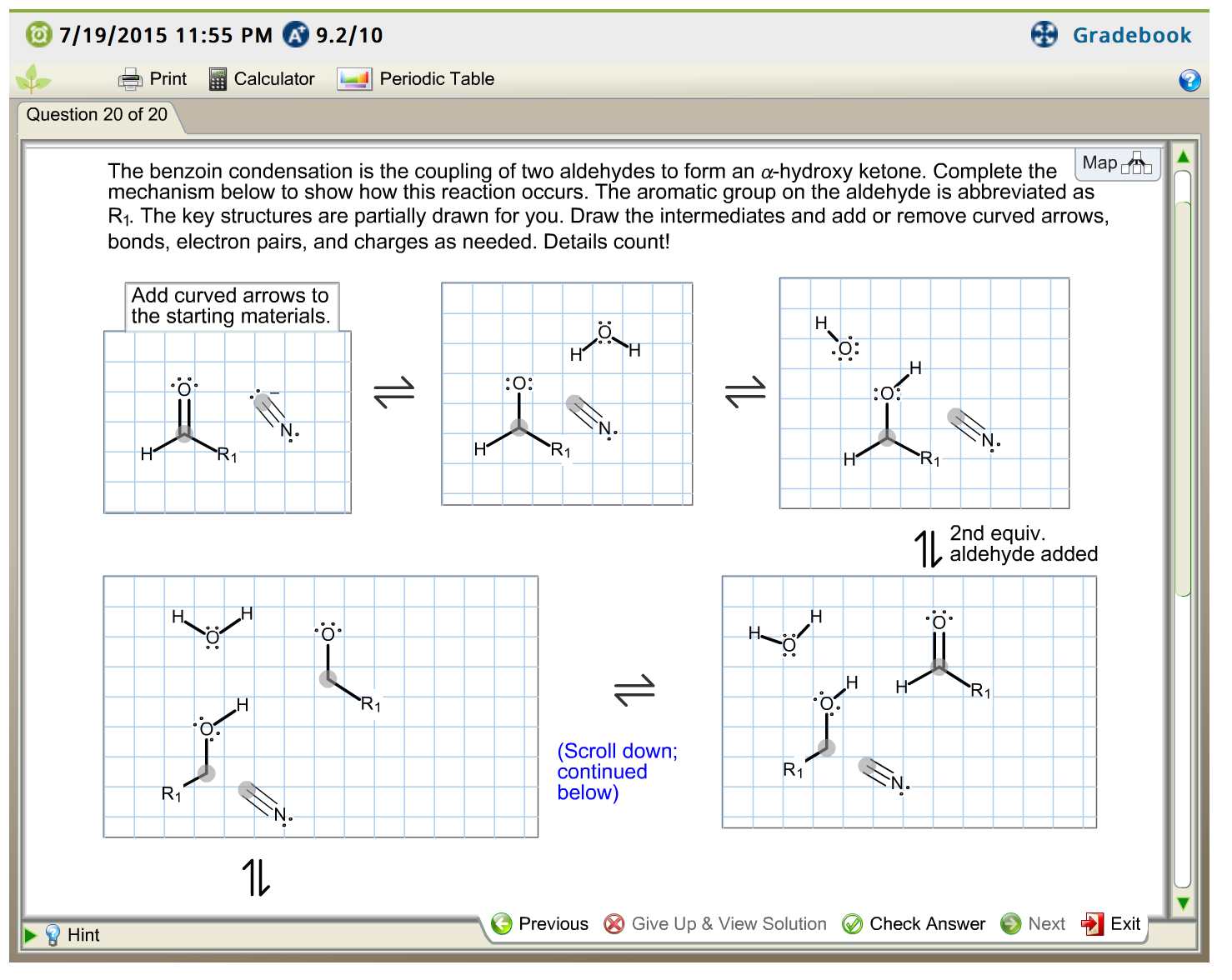
Solved The benzoin condensation is the coupling of two
Oxidation of alcohols to aldehydes is partial oxidation; Web the oxidation of an alcohol to form an aldehyde or ketone is very important in synthesis. Web answer 1 aldehydes are a class of organic compound which can be oxidized to form corresponding carboxylic acid or organic acid. Web aldehydes, rcho, can be oxidised to carboxylic acids, rco2h. O c h.
They Use A Strong Oxidant Like Potassium Permanganate (Kmno4)
Web the product of the oxidation of an aldehyde, results in a carboxylic acid. Oxidation of alcohols to aldehydes is partial oxidation; Web answer 1 aldehydes are a class of organic compound which can be oxidized to form corresponding carboxylic acid or organic acid. Web aldehydes, rcho, can be oxidised to carboxylic acids, rco2h.
Web The Oxidation Of Aldehydes By O2 Appears To Be A Chain Mechanism Involving Free Radicals And Yielding A Rather Reactive Peracid, Which Then Reacts With The.
Web there are some reagents which can selectively oxidize a primary alcohol and stop at an aldehyde without complete oxidation to the acid. Aldehydes are further oxidized to carboxylic acids. Web oxidation of 1 o alcohols to form aldehydes (section 17.7) pcc pyridinium chlorochromate ( pcc) is a milder version of chromic acid. In this process, the hydroxy hydrogen of the alcohol is replaced by a leaving group (x in.
Web Aldehydes Undergo Oxidation More Quickly Than Ketones.
Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids. Ketones are not oxidised under these conditions as they lack the critical h for the elimination to occur (see. Primary alcohols can only be oxidized to form aldehydes or carboxylic acids.for instance, using chromic acid (to get a carboxylic acid), or using pcc in. In this case, excess dichromate will further oxidize the aldehyde to a.
O C H O C O H Oxidation ¥Alcohols.
This will happen if the oxidation happens under acidic or alkaline conditions. These functional groups are useful for further reactions; Web oxidation of alcohols to aldehydes and ketones. Web currently, most investigations on aldehyde oxidations focus on aerobic oxidation, i.e., using molecular oxygen (o 2) to oxidize aldehydes into the corresponding carboxylic.